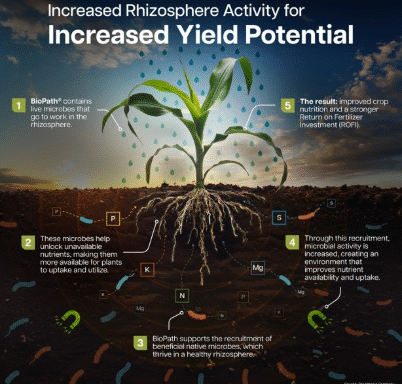
Winter Park, Florida–(Newsfile Corp. – December 15, 2025) – Adia Nutrition Inc. (OTCQB: ADIA), a leader in regenerative medicine through its Adia Med subsidiary, today announced that the independent Institutional Review Board (IRB) BeyondBound has approved the company’s patent-pending clinical study protocol focused on Autism Spectrum Disorder (ASD).IRB Approval EmailTo view an enhanced version of this graphic, please…