The FDA has issued another complete response letter to Regeneron over its high-dose Eylea due to unresolved issues at a Novo Nordisk manufacturing plant. The company, however, is making progress in lining up alternate manufacturing help.
Regeneron reports another manufacturing-related rejection for Eylea HD, but says help is on the way
Latest from Blog

Nutrition nonprofit holds roundtable discussion in Fitzgerald
While their focus may be on providing aid around the globe, MANA Nutrition is doing what they can to keep American farmers in mind. Post Views: 2
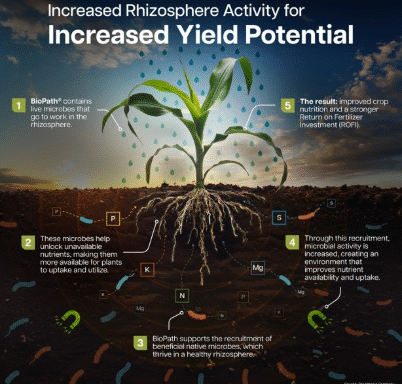
ROFI Drives New Approach to Crop Nutrition
With input costs continuing to pressure farm budgets, some growers are making difficult decisions to reduce fertilizer application rates. While that strategy may ease upfront expenses, it is also accelerating a broader
Tippecanoe schools hire new nutrition leader
Alison Jordan has been hired as the new nutrition services director for Tippecanoe School Corporation, overseeing meals for 20 schools. Post Views: 2

Gift Nets Scholarships, Nutrition for Men’s Soccer
The $110,000 gift comes from the New Hope Foundation, which works to create opportunities for individuals and families in need. Post Views: 1

Adia clears ethics review for CKD stem cell trial, recruitment next
The planned trial targets adults with Stage 2-4 chronic kidney disease and will track kidney function and inflammation markers. Recruitment starts after listing. Post Views: 1